What Do Major Guidelines Say About the Oncotype DX® Assay?
The Oncotype DX assay is the only multigene breast cancer assay incorporated into four major clinical guidelines: those from the St Gallen Consensus guidelines, the European Society of Medical Oncology, National Comprehensive Cancer Network® (NCCN®), and the American Society of Clinical Oncology® (ASCO®). All four have included the Oncotype DX breast cancer assay in their guidelines as an option to predict whether certain patients will benefit from chemotherapy.
St Gallen
The 13th St Gallen International Breast Cancer Conference Expert Panel, for the second time, recognized the Oncotype DX breast cancer assay for its ability to provide not only prognostic, but also predictive information regarding the utility of cytotoxic therapy in addition to endocrine therapy for patients with node-negative or node-positive early stage invasive breast cancer. The Panel considered that only the 21-gene RS was predictive of chemotherapy responsiveness, though a substantial minority would also endorse PAM50 or the 70-gene signature for this purpose. This led to a recommendation that selection of patients who might forego chemotherapy could be based on the 21-gene RS, but the Panel did not offer majority endorsement for PAM50, the 70-gene signature or EPClin as yet established for this purpose
Reference: Goldhirsch A, et al. Ann Oncol 2013; 24:2206–2223. St Gallen International Consensus panel does not endorse any product or therapy.
European Society of Medical Oncology (ESMO)
Gene expression profiles such as Oncotype DX Recurrence Score® result (Genomic Health, Redwood City, USA) may be used to gain additional prognostic and/or predictive information to complement pathology assessment and to predict response to adjuvant chemotherapy.
Reference: Senkus E, et al. Ann Oncol 2013;24: Suppl 6:vi7-23.
NCCN®
"On retrospective analysis of two trials (NSABP B-14 and B-20) performed in women with hormone receptor-positive, axillary lymph node-negative invasive breast cancer, this assay system was able to quantify risk of recurrence as a continuous variable (eg, Oncotype DX Recurrence Score) and to predict responsiveness to both tamoxifen and CMF or methotrexate/5-fluorouracil/leucovorin chemotherapy."
NCCN Clinical Practice Guidelines in Oncology™: Breast Cancer (Version 2.2011)
ASCO®
The Oncotype DX assay may be used in newly diagnosed ER+, N- invasive breast cancer patients to predict risk of recurrence. The assay can also be used to identify patients who may be successfully treated with tamoxifen and may not require adjuvant chemotherapy. "It has been suggested that tamoxifen-treated patients with an excellent estimated prognosis may be spared adjuvant chemotherapy." Access these guidelines.
Harris L, Fritsche H, Mennel R, et al. American Society of Clinical Oncology 2007 update of recommendations for the use of tumor markers in breast cancer. J Clin Oncol. 2007;25:5287-5312.
The UK's National Institute for Health and Care Excellence (NICE) recommended the Oncotype DX assay for use in clinical practice to guide adjuvant chemotherapy treatment decisions for certain patients with early-stage breast cancer, including micrometastatic disease. Access this guidance.
Insight Beyond Traditional Markers
The Oncotype DX Recurrence Score results provide additional insight into underlying tumour biology beyond traditional measures such as patient age, tumour size, tumour grade, Ki-67, and ER status 1-10
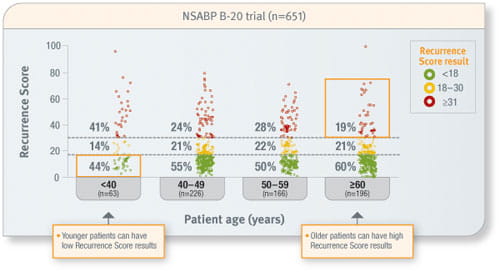
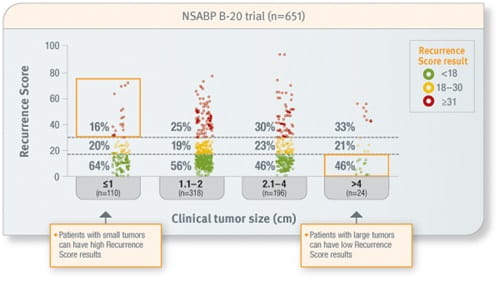
Patient age and tumour size cannot predict the Recurrence Score result
Tumour size cannot predict Recurrence Score result
NSABP B-20
Prospective analysis of archived tissue from 651 patients with ER-positive, node-negative invasive breast cancer treated with tamoxifen or tamoxifen plus CMF/MF. Approximately 45% of the patients were <50 years of age, two-thirds of tumours were ≤2.0 cm in size, and 20% of tumours were PR-negative.
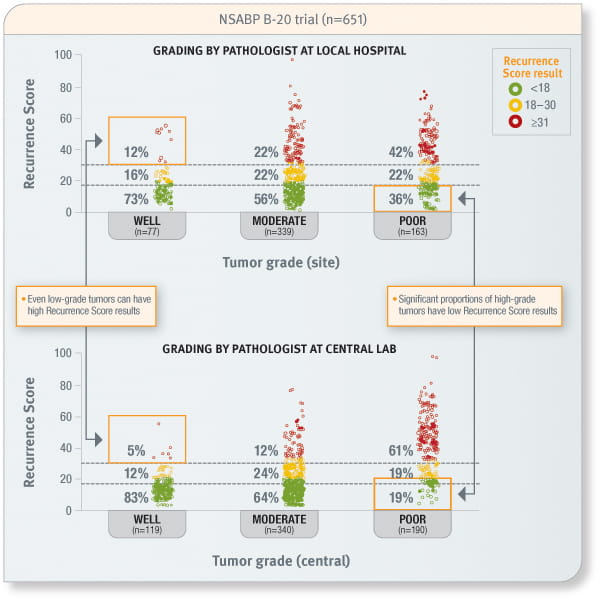
Tumour grade cannot predict the Recurrence Score result
The Oncotype DX assay reveals critical information that changes treatment decisions.1
Because high and low Oncotype DX Recurrence Score results reflect different intrinsic tumour biology, physicians can make decisions based on underlying disease.
Combinations of patient age, grade, and tumour size cannot predict the Recurrence Score result1
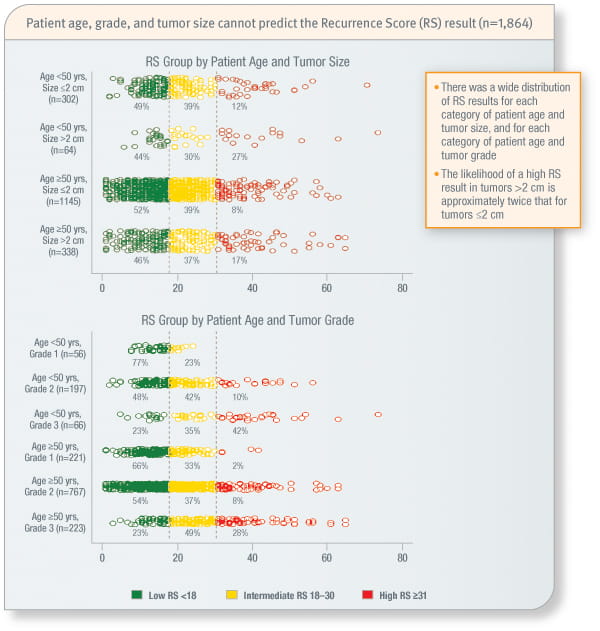
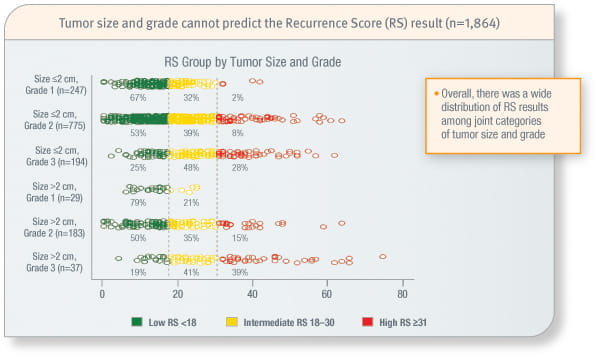
Study included 1,864 ER-positive, HER2-negative, node-negative patients from the Clalit and Maccabi Health Services in Israel.
Oncotype DX provides an individualised Recurrence Score result that cannot be predicted by traditional clinicopathologic variables alone or in combination.1
Ki-67 cannot predict predict the Recurrence Score result6
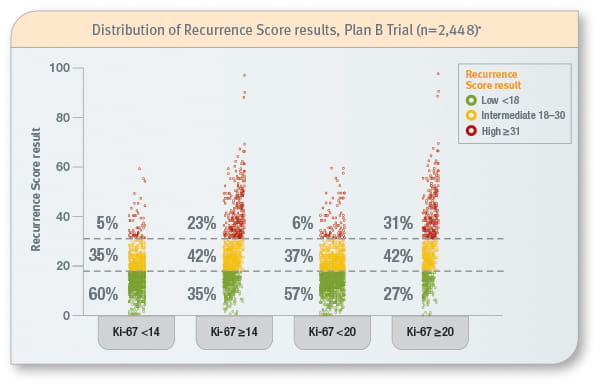
* The Phase III West German Study Group (WSG) Plan B Trial prospectively evaluated the Recurrence Score results in 2,448 patients with hormone receptor-positive early-stage invasive breast cancer. Ki-67 assessment was done by IHC through central pathology.
- There is a wide range of Recurrence Score results for varying Ki-67 cutoffs. There is only moderate correlation between the Recurrence Score result and Ki-67 (rs = 0.374; p<0.001)6,7
- The Recurrence Score result cannot be predicted by Ki-67
- Currently the clinical utility of Ki-67 in early-stage invasive breast cancer is limited due to lack of analytic reproducibility and standardization8
Intrinsic subtypes
Quantitative RT-PCR provides insight beyond traditional measures9,10
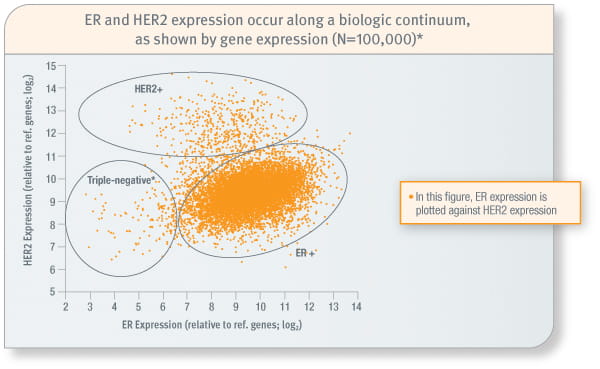
*More than 99% of these cases were progesterone receptor–negative; rarely strongly progesterone receptor–positive.
Study included 100,000 invasive breast cancer tumour specimens that were examined in the Genomic Health laboratory from July 2005 through May 2009. Quantitative expression for each gene was measured by the Oncotype DX assay on a scale from 0 to 15, relative to reference genes.
- Quantitative RT-PCR is able to identify a continuum of gene expression10
- Three groups are discernible: HER2-positive, ER-positive/HER2-negative, and a triple-negative group10
- Within each group is a wide distribution of expression and heterogeneity that might not be detected by binary methods
In a multivariate analysis of clinicopathologic measures from NSABP B-20, the Oncotype DX Recurrence Score result was the strongest predictor of chemotherapy benefit in node-negative patients1
REFERENCES
1. Paik S, Tang G, Shak S, et al. Gene expression and benefit of chemotherapy in women with node-negative, oestrogen receptor–positive breast cancer. J Clin Oncol. 2006;24:3726-3734.
2. Paik S, Shak S, Tang G, et al. A multigene assay to predict recurrence of tamoxifen-treated, node-negative breast cancer. N Engl J Med. 2004;351:2817-2826.
3. Habel L, Shak S, Jacobs M, et al. A population-based study of tumour gene expression and risk of breast cancer death among lymph node-negative patients. Breast Cancer Res. 2006;8:R25-R39.
4. Shak S, Baehner F, Stein M, et al. Quantitative gene expression analysis in a large cohort of oestrogen-receptor positive breast cancers: characterisation of the tumour profiles in younger patients (≤40 years) and in older patients (≥≥70 years). Presented at: 33rd Annual San Antonio Breast Cancer Symposium; December 8-12, 2010; San Antonio, TX. Poster P3-10-01.
5. Liebermann N, Baehner FL, Soussan-Gutman L. Evaluation of Recurrence Score and traditional clinicopathologic assessments in a large oestrogen receptor-positive, lymph node-negative patient cohort. [ASCO abstract 632]. J Clin Oncol. 2011;29(suppl).
6. Abstract #552. Gluz O, Kreipe HH, Christgen M, et al. Prospective comparison of recurrence score and independent central pathology assessment of prognostic tools in early breast cancer (BC): focus on HER2, ER, PR, Ki-67 results from the phase III WSG-Plan B trial. Poster presented at: American Society for Clinical Oncology Annual Meeting; June 2012; Chicago, IL.
7. Data on File. Genomic Health, Inc. Redwood City, CA.
8. Harris L, Fritsche H, Mennel R, et al. American Society of Clinical Oncology 2007 update of recommendations for the use of tumour markers in breast cancer. J Clin Oncol. 2007;25(33):5287-5312.
9. Baehner F, Watson D, Anderson J, et al. Quantitative gene expression by RT-PCR in the special histologic subtypes of oestrogen receptor positive invasive breast cancer. Presented at: 99th Annual Meeting of United States & Canadian Academy of Pathology; March 20-26, 2010; Washington, DC. Abstract 150.
10. Shak S, Baehner F, Palmer G, Ballard J, Baker J, Watson D. Subtypes of breast cancer defined by standardised quantitative RT-PCR analysis of 10,618 tumours. Presented at: 29th Annual San Antonio Breast Cancer Symposium; December 14-18, 2006; San Antonio, TX. Abstract 6118.
NCCN is a registered trademark of the National Comprehensive Cancer Network; ASCO is a registered trademark of the American Society of Clinical Oncology; ESMO is a registered trademark of European Society of Medical Oncology. NICE, St. Gallen, ESMO, NCCN and ASCO do not endorse any therapy or product.
