Gain foresight using genomic insights
The Colon Recurrence Score® test results provide a precise estimate of the risk of recurrence for anatomic stage II, MMR-P, or stage III A/B patients to help guide the most appropriate treatment decision.1-4
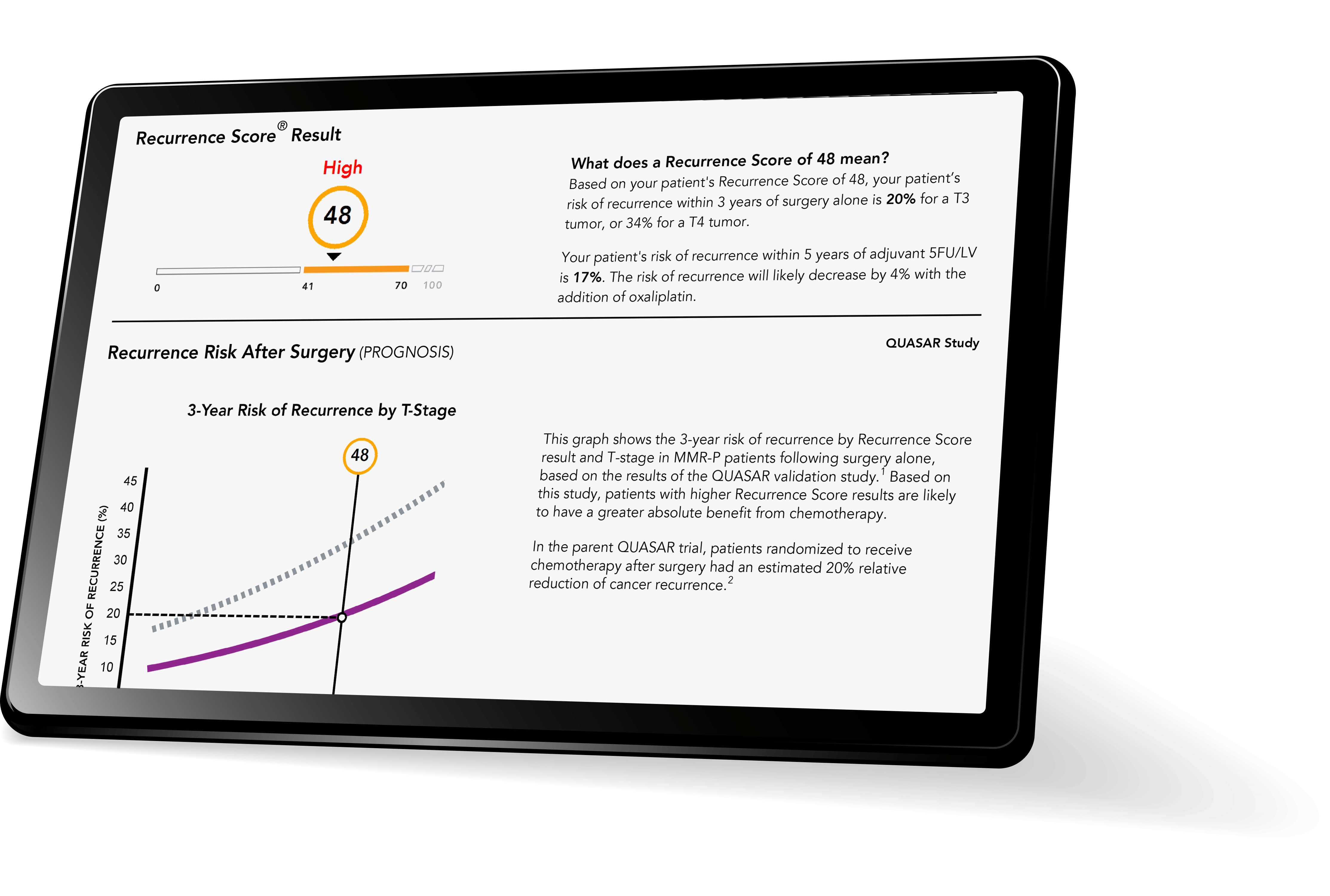
High Recurrence Score® Result
Identifies anatomic stage II MMR-P patients at higher risk of recurrence.1-4
These patients may have a larger absolute benefit from chemotherapy
Low Recurrence Score Result
Identifies anatomic stage III A/B patients at lower-than-average risk of recurrence.3,4
These patients may have a smaller absolute benefit from the addition of oxaliplatin to fluorouracil and leucovorin (5-FU/LV).
For your Stage II MMR-Proficient, T3 or T4, and Stage III A/B colon cancer patients, it’s time to consider the Colon Recurrence Score test.
A genomic test for colon cancer with level 1 evidence
The Colon Recurrence Score test is backed by data developed over the course of multiple collaborative studies with both anatomic stage II and stage III patients.
Four validation studies—QUASAR, CALGB 9581, NSABP C-07, and SUNRISE—confirm that the Colon Recurrence Score test result predicts the risk of local recurrence.1-4 In addition, studies show that physicians often change treatment recommendations based on the test results.5-8
Genes that provide real insights into colon cancer tumor biology
The Colon Recurrence Score test was developed in 4 steps resulting in a 12-gene panel and algorithm that’s been validated in several studies.1-4,9-11

Optimization of methods for quantifying gene expression in formalin-fixed, paraffin-embedded tissue (FPET).

Selection of 761 candidate genes from the human genome.

Testing of candidate genes to identify an optimal gene panel for clinical validation.

Prospective clinical validation of the 12-gene panel and Colon Recurrence Score result calculation.
The Oncotype DX Colon Recurrence Score 12-Gene Panel
| Cell Cycle | Stromal |
ATPSE PGK1 GPX1 UBB VDAC2 |
|
Ki-67 C-MYC MYBL2 |
FAP BGN INHBA |
|
| GADD45B | ||
| 7 cancer genes consistently associated with colon cancer recurrence | 5 reference genes normalize the expression of cancer-related genes | |
Is your patient eligible?
The Oncotype DX Colon Recurrence Score test is for patients who:
- Are newly diagnosed with anatomic stage II, T3 or T4, MMR-P, or stage III A/B colon cancer
- Have undergone surgical resection
- Have not started chemotherapy or other treatment
Order a test
We make your ordering experience efficient. You can order tests through the physician portal, and we’ll help you every step of the way.
Coverage and financial assistance
The Colon Recurrence Score test is widely accessible with minimal out-of-pocket costs for most patients.*
*The Oncotype DX Colon Recurrence Score test is performed on claims with the unique proprietary laboratory analysis (PLA) code, 81525.
-
References

